Participant Recruitment & Screening

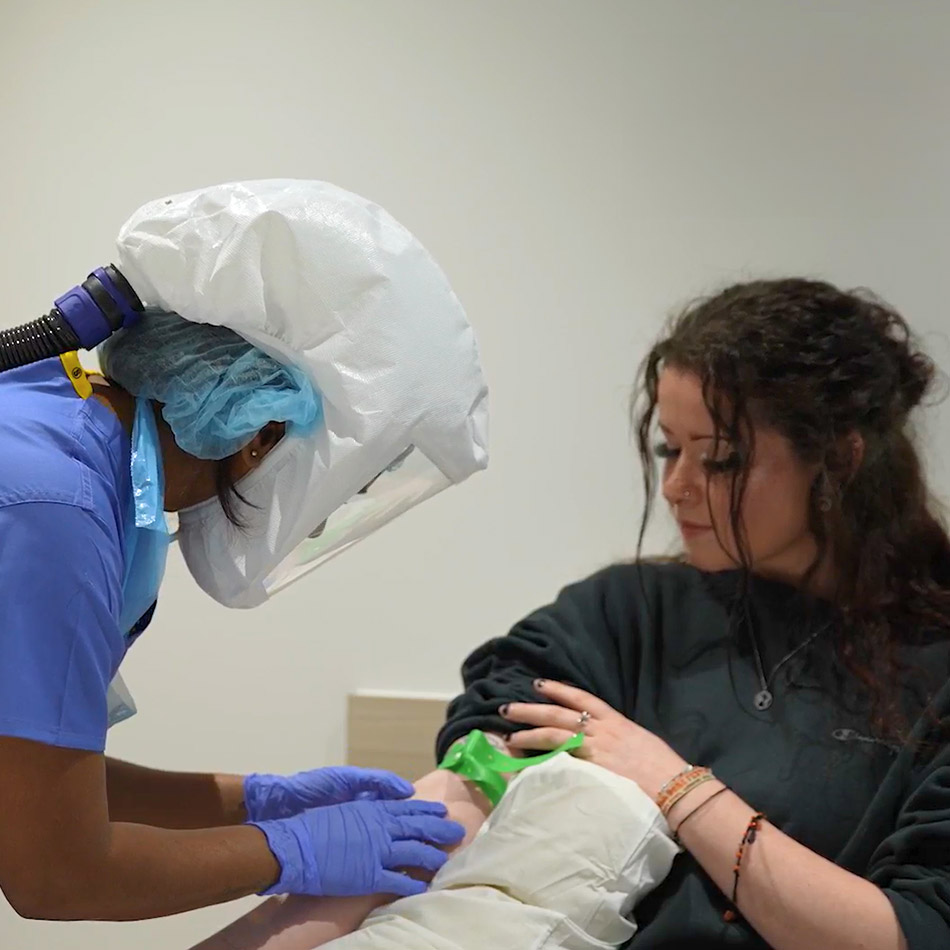
hVIVO recruits, screens and selects healthy volunteers for human challenge studies using robust ethical, scientific and medical frameworks. Our recruitment and screening pathway ensures participant safety while providing high‑quality, well‑characterised study cohorts.
Reliable and rapid participant recruitment and screening is the catalyst for full cohorts required for all human challenge studies. We identify individuals who meet the strict medical, demographic and immunological criteria required for controlled viral challenge. This includes outreach, pre‑screening, clinical assessments, informed consent, laboratory testing, and cohort allocation — all conducted under rigorous ethical, GCP and regulatory requirements.
400,000+
participants in our FluCamp database
Purpose‑built screening clinic in London
PI‑led medical oversight throughout eligibility assessment
In house recruitment team
How We Work
- Targeted Recruitment Campaigns via FluCamp database
- Pre‑Screening Questionnaire
- In‑Clinic Screening Visit
- Challenge‑Model–Specific Immunological Screening
- Final Medical Review & Eligibility Confirmation
- Cohort Allocation & Study Preparation
Our Capabilities
- Large volunteer database (>400,000 opted‑in participants)
- Dedicated screening facilities in London
- Immunology & serology testing via our bioanalytical labs
- Viral challenge–specific eligibility assays
- GCP‑aligned clinical processes
- Workforce of specialist screening nurses and medical officers
- Real‑time scheduling and participant management system
- Multilingual recruitment capability


Why hVIVO?
- Unmatched challenge study expertise
- High‑quality cohorts
- End‑to‑end recruitment infrastructure
- Speed and predictability
- Participant safety first
Evidence
-
Rapid recruitment of 200+ participants for a multi-cohort influenza challenge study
-
Successfully enrolled hard‑to‑reach cohorts via targeted digital outreach.
-
Reduced screening‑to‑enrolment timelines by 30%.
FAQ
Through our FluCamp volunteer platform, targeted digital campaigns, and our large opted‑in database.
Bring your challenge model to life with expert scientific support
