Clinical Trial Supplies & Kit Production
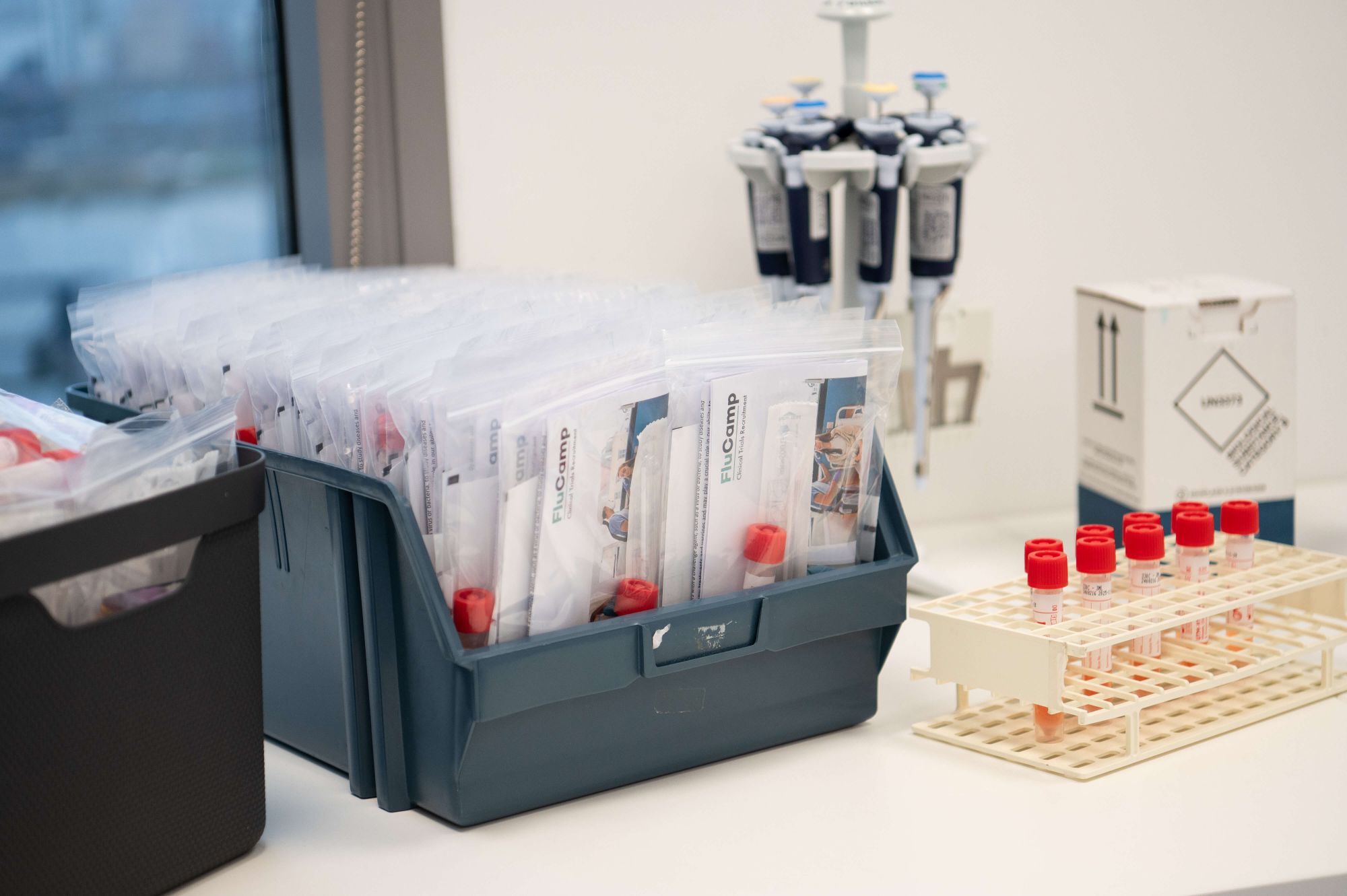
hVIVO Supplies & Kit Production
How hVIVO Solves your Clinical Trial Supply and Kit Production Challenges
-
Lab manual production
-
Site training
-
Investigator Support: Comprehensive assistance for investigators to ensure protocol adherence and smooth site operations
-
Bespoke kit design to meet your specific requirements, customisation for complex protocols
-
Scalable batches for your study requirements
-
Kit production in house - aligned with laboratory and protocol requirements
-
hVIVO’s RSV stabilisation matrix with extended stability maintaining virus infectivity, securing data collection
-
Global, multi-site distribution and temperature-controlled shipping, LIMs barcoded
Phase Coverage
-
Support for Phase I–III clinical trials with tailored solutions for each stage.
We provide end‑to‑end clinical trial supply and kit production services, including custom kit assembly, labelling, packaging, storage, and distribution. Our solutions support a wide range of study designs, from early‑phase to late‑phase trials, and can be tailored to protocol‑specific and investigator requirements.
Research & Articles
A New Frontier in Respiratory Health: Next-Gen Human Challenges Explained
Advancing Respiratory Virus Research Through Next‑Generation Human Challenge Models
No articles found for this topic.
Plan your next Clinical Trial with hVIVO
If you're a potential customer, find out how hVIVO can help you.
