The Challenge
RSV Human Challenge Models
Healthy participants 18-55 year old
Older Adults aged 60-75 years old
Conducted a number of human RSV challenge studies for a range of customers:
- Currently, the only RSV challenge model commercially available
- Healthy participant model utilised across multiple studies with vaccines and antivirals directed towards RSV
- RSV challenge model available in Older Adults (60-75) – targets population associated with significant unmet need in RSV
As the industry leader in conducting human viral challenge studies, we have developed a large Virobase, of clinical data paired with virological data, host genetics and immunology combined with an extensive biorepository of blood and respiratory samples. This “Virometrics” resource, in conjunction with our unique insight into the host response to viral disease, allows us to tailor study designs to each IMP.
"Congratulations Team!! Well done and thank you all for your incredible efforts"
US Biotech client
Vaccines
Conceptual Challenges:
- Testing proof of concept in the target population (pediatrics or elderly)
- No clear product development pathway
- Correlates of infection are poorly understood
- Lack of fully translatable immunogenicity animal models
Our Human Challenge Models: Towards a deeper understanding
- Healthy adult model enables fast efficient proof of concept demonstration and safety in disease model before moving to at risk populations in field trials
- Elderly (60+) model, enables demonstration of proof of concept in target population with weakened immunity
- For vaccines, both options facilitate efficient investigation of known and novel correlates of vaccine protective effect

Antivirals/Treatments
Conceptual Challenges:
- Testing proof of concept in the target population (pediatrics or elderly)
- Correlates of infection are poorly understood
- Lack of fully translatable immunogenicity animal models
Our Human Challenge Models: Towards a deeper understanding
Study design matched to investigational product mechanism of actions.
- Optimisation of treatment timing
- Time-dependent measurements of biomarkers
- Triggered-dosing options (time or virological)
- Controlled strain exposure
- Consistent placebo response
- Efficient resistance monitoring

Immunomodulators
Conceptual Challenges:
- Limited RSV immunomodulators in development
- Limited focus on adult/elderly populations
Our Human Challenge Models: Towards a deeper understanding
- Well controlled quarantine environments
- Baseline well established prior to infection
- Appropriate for both prophylaxis and treatment
- Flexible dosing and timing
- Establish safety & efficacy to impact infected subjects host response
- Investigate and demonstrate target engagement
- Controlled combination-treatment with drug and standard of care
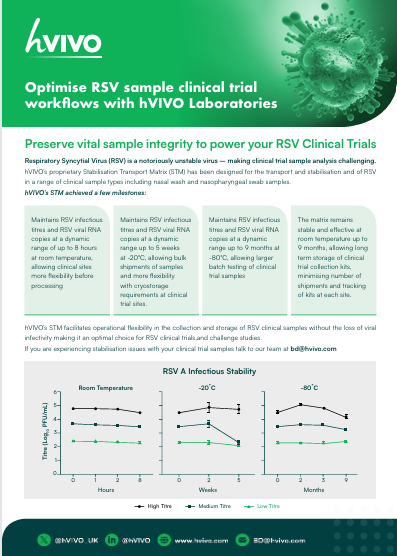
Optimise RSV Sample Workflows
hVIVO’s proprietary Stabilisation Transport Matrix (STM) has been designed for the transport and stabilisation and of RSV in a range of clinical sample types including nasal wash and nasopharyngeal swab samples
Related Resources
Real‑World RSV Maternal Vaccine Data Marks a Turning Point for Infant Protection

A Landmark Moment for Pertussis Vaccine Development — and for the Future of Bacterial Challenge Models

Fast‑Tracking First‑in‑Human: Five Lessons Startups Can’t Afford to Miss

The Early‑Phase Anchor: Why Medical Monitoring Matters Long Before the First Dose

Paper: Mucosal and systemic immune correlates of viral control after SARS-CoV-2 infection challenge in seronegative adults

From RSV to hMPV: Building the Next Generation of Respiratory Challenge Models

Why are Biotech’s & Pharma Using Human Challenge Studies for Drug Development?

Tripledemic Takedown: How Human Challenge Trials are Ideal to Expedite Multivalent Vaccine Development

Empowering Next‑Gen Infectious Disease & Vaccine Development
Human Challenge Studies: Their Conduct and Safety Aspects

Bring your challenge model to life with expert scientific support
