Microbiology Laboratory Services
Schedule a Call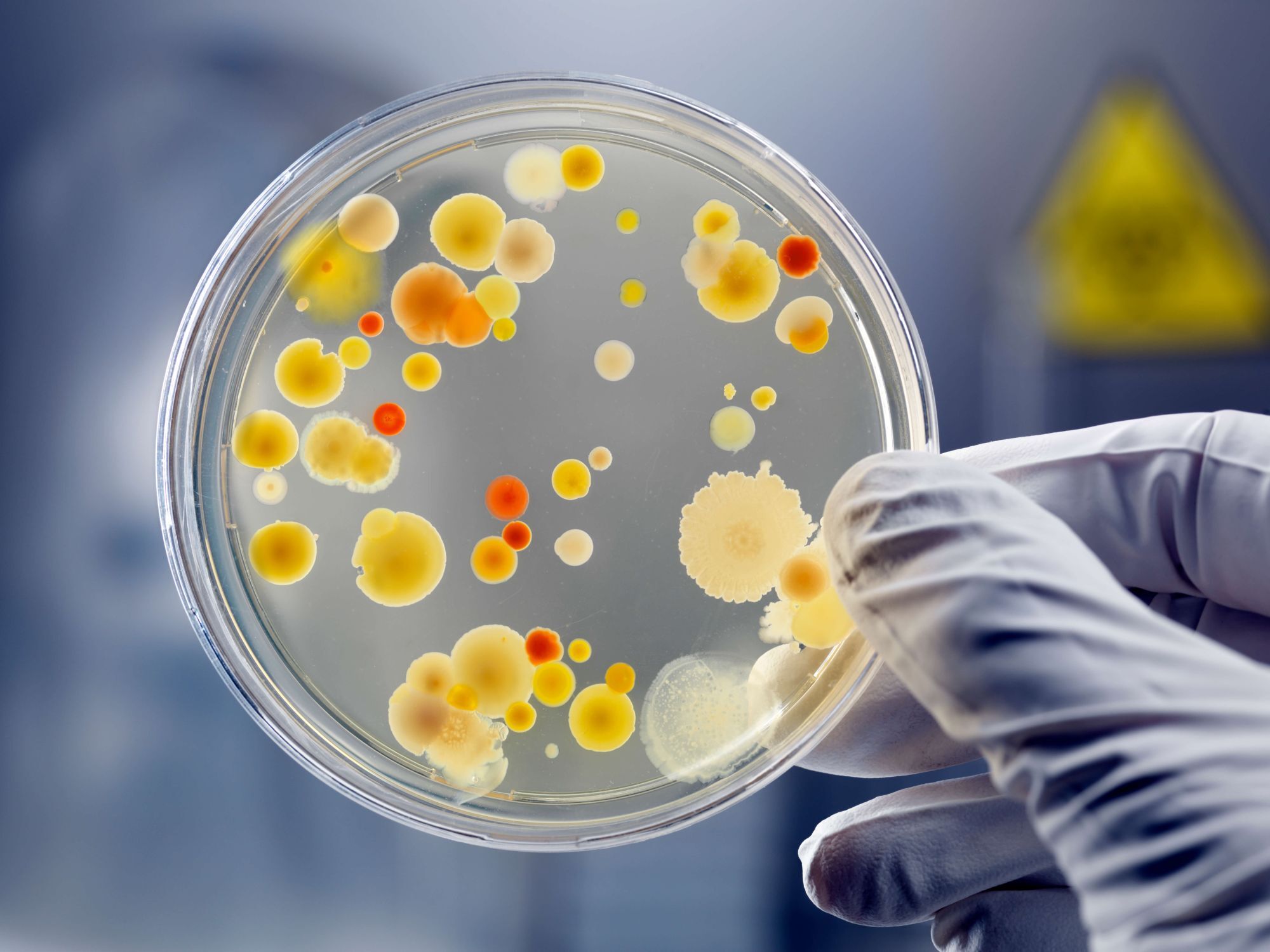
Microbial detection and characterisation for infectious disease research
Our Services
-
Microbiome sequencing using NGS
-
Culture-based microbial identification
-
Molecular detection of pathogens
-
Antimicrobial susceptibility testing
Applications
-
Infectious disease research
-
Gut microbiome studies
-
Antimicrobial resistance monitoring

Assays as Architecture: Building Blocks for Translational Research
Providing structure and validity When people think about assays, they often picture routine lab tests — a box to tick, a dataset to generate. But in our world, assays are much more than that. They are the building blocks, the architecture that supports translational research from the ground up. Without them, studies lack structure and validity. With them, we can design programmes that are robust, scalable and ready to expand into new therapeutic areas. Why assays must be planned early Assays are not add‑ons, they are the means by which to convert biological processes to meaningful and measurable data. They need to be considered at the very beginning of study design. When assays are built into the framework early, they shape the analytical strategy downstream. They provide consistency across phases of development, from pre‑clinical through to late‑stage clinical, and they give sponsors confidence that the data they generate will stand the test of time. A one‑off test might answer a short‑term question, but a validated assay becomes infrastructure — something that can support a programme for years. Expertise rooted in infectious disease At hVIVO, our laboratory expertise was perfected in infectious and respiratory disease. We built platforms to support human challenge studies, vaccine development and antiviral testing. That meant investing in molecular biology, immunology and microbiology capabilities, and creating teams dedicated to assay development and validation. Over time, those building blocks became a foundation we could use again and again, not just for one project but across a portfolio of studies. Expanding the architecture Today, that foundation is expanding. We have added digital PCR, Ligand binding assay diversity, multiplexing and next‑generation sequencing, robotics and automation, as well as expanding capabilities in microbiology and cell culture. These platforms allow us to work with smaller sample volumes, higher throughput and greater flexibility. They also open doors to new indications: oncology, metabolic disease, cardiovascular research and even gene therapy. The same architectural approach applies — build the assays, validate them, and then use them as infrastructure to support long‑term development. From infectious disease to broader translational science What excites us is the agility this gives sponsors. We can combine platforms, tailor assays to specific development stages and adapt to new therapeutic areas. Whether it is neutralising antibody assessments for vaccines, biomarker panels for oncology or microbial propagation for antibacterial programmes, the principle is the same: assays are the structure that holds the science together. Conclusion Assays are not just tests. They are architecture. They are the foundations on which translational research is built, the framework that allows innovation to scale across indications and phases. By treating assays as building blocks, we create infrastructure that supports sponsors from early discovery through to clinical development — and ultimately helps bring better therapies to patients.
Related Resources
Empowering Next‑Gen Infectious Disease & Vaccine Development
Enhancing Our Full-Service Offering: hVIVO’s New Bacteriology Lab

Advantages to Centralised Laboratory Services

Assays as Architecture: Building Blocks for Translational Research
Why NGS Is the Missing Link in Early‑Phase Infectious Disease Research

Controlled Human Infection Model's (CHIM) in your backpack: how Human Challenge Trials support regulatory approval of travel vaccines

Fact Sheet: De-Risking Sample Management
Partner with hVIVO’s expert Laboratory Teams to deliver high-quality Clinical Data
Speak to our team about your study requirements.
